Research Projects
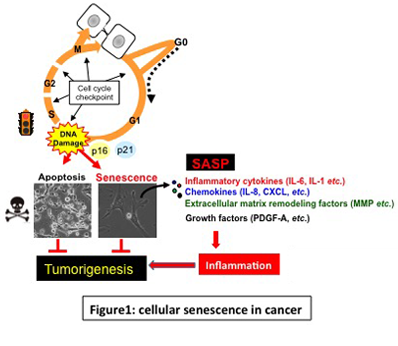
Cellular senescence is the state of irreversible cell cycle arrest that can be induced by a variety of potentially oncogenic stimuli and has therefore long been considered to suppress tumorigenesis, acting as a guardian of homeostasis. Emerging evidence, however, reveals that senescent cells also promote secretion of various inflammatory and pro-proliferative factors. This newly identified senescence-associated phenotype termed SASP is likely to be associated with homeostatic disorders including cancer. It is therefore quite possible that accumulation of senescent cells during aging or obesity in vivo may contribute to aging- and/or obesity-associated cancers. By conducting the following studies, we aim to clarify the molecular mechanisms underlying aging- and/or obesity-associated cancer.
1. Research aimed at elucidating the mechanism by which senescent cells increase in the body with aging and exploring methods to control this increase.
2. Research to elucidate the relationship between cellular senescence and SARS-CoV-2 infection and its sequelae.
3. Research to identify gut bacteria that induce cellular senescence and to elucidate their relationship to colorectal cancer.
4. Research aimed at developing a method to specifically remove senescent cells from the body.
5. Research aimed at elucidating the induction mechanism of SASP and its control.