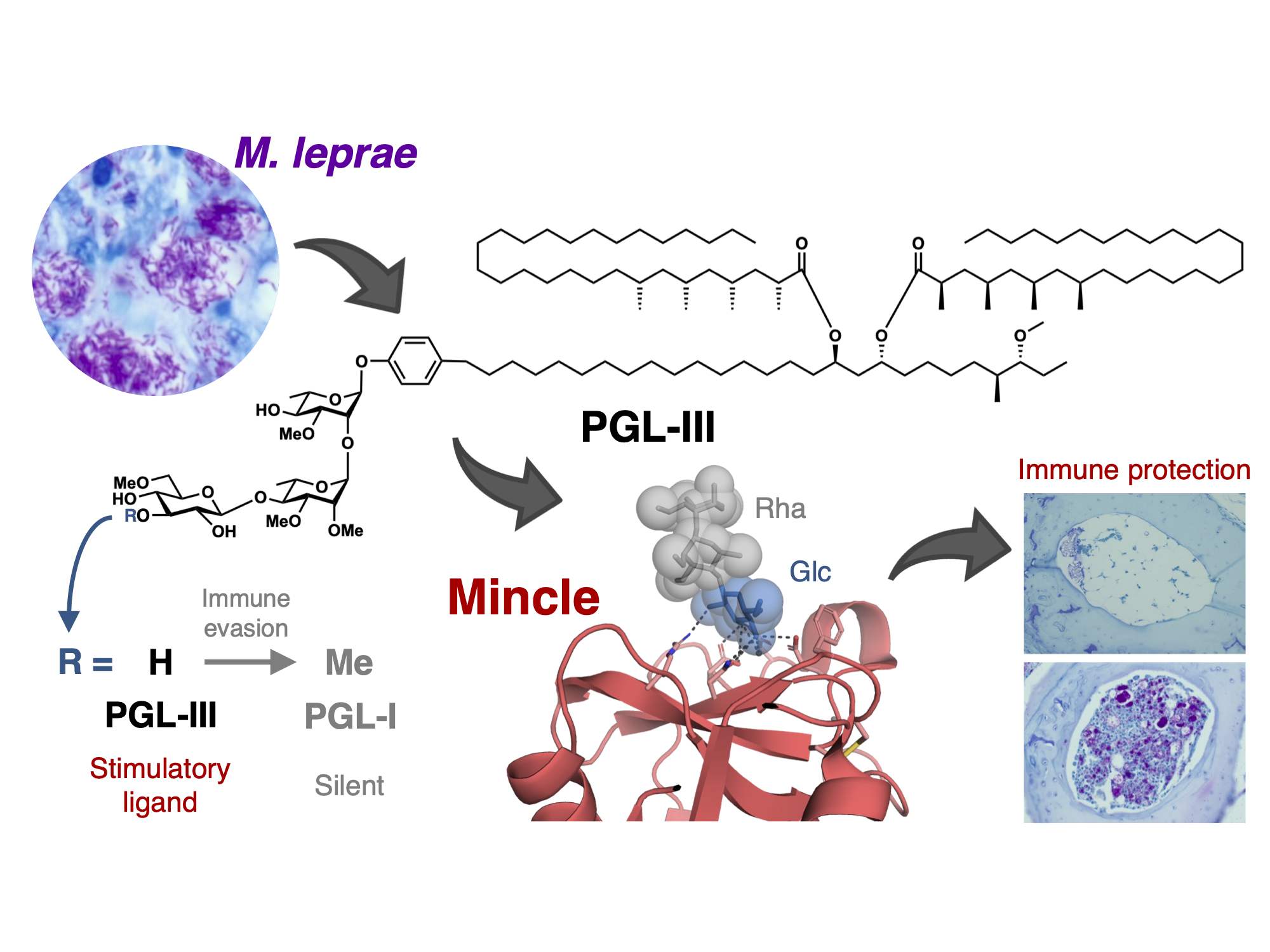
Discovery of the mechanism by which the leprosy bacillus disrupts macrophages (Yamasaki Lab, in ACS Central Science)
Although leprosy (Hansen’s disease) is one of the oldest known diseases, the pathogenicity of Mycobacterium leprae (M. leprae) remains enigmatic. Indeed, the cell wall components responsible for the immune response against M. leprae are as yet largely unidentified. We here reveal phenolic glycolipid-III (PGL-III) as a M. leprae-specific ligand for the immune receptor Mincle. PGL-III is a scarcely present trisaccharide intermediate in the biosynthetic pathway to PGL-I, an abundant and characteristic M. leprae glycolipid. Using activity-based purification, we identified PGL-III as a Mincle ligand, that is more potent than the well-known M. tuberculosis trehalose dimycolate. The co-crystal structure of Mincle and a synthetic PGL-III analogue revealed a unique recognition mode, implying that it can engage multiple Mincle molecules. In Mincle-deficient mice infected with M. leprae, increased bacterial burden with gross pathologies were observed. These results show that PGL-III is a non-canonical ligand recognized by Mincle triggering protective immunity.
This article is published in ACS Central Science on July 12, 2023.
Title: “PGL-III, a rare intermediate of Mycobacterium leprae phenolic glycolipid biosynthesis, is a potent Mincle ligand”
Authors: Shigenari Ishizuka#, J. Hessel M. van Dijk#, Tomomi Kawakita, Yuji Miyamoto, Yumi Maeda, Masamichi Goto, Guillaume Le Calvez, L. Melanie Groot, Martin D. Witte, Adriaan J. Minnaard, Gijsbert A. van der Marel, Manabu Ato, Masamichi Nagae, Jeroen D. C. Codée, and Sho Yamasaki (#Equally contributed)
Links
- Home
- Achievement
- Research Activities
- Discovery of the mechanism by which the leprosy bacillus disrupts macrophages (Yamasaki Lab, in ACS Central Science)