Cellular senescence-associated factors in MDSCs promote tumour growth (Hara lab in Nat. Commun)
Cellular senescence is a state of irreversible cell cycle arrest and induced by oncogenic stress. Cyclin-dependent kinase inhibitors (CDKIs) p16Ink4a and p21Cip1/Waf1 play an important role in tumour suppression through induction of cellular senescence. Indeed, mice lacking p16Ink4a and/or p21Waf1/Cip1 exhibit early onset of cancer and we previously revealed the roles of p16Ink4a and/or p21Waf1/Cip1 expression during tumour formation in vivo. However, senescence-independent roles of these CDKIs are not well understood.
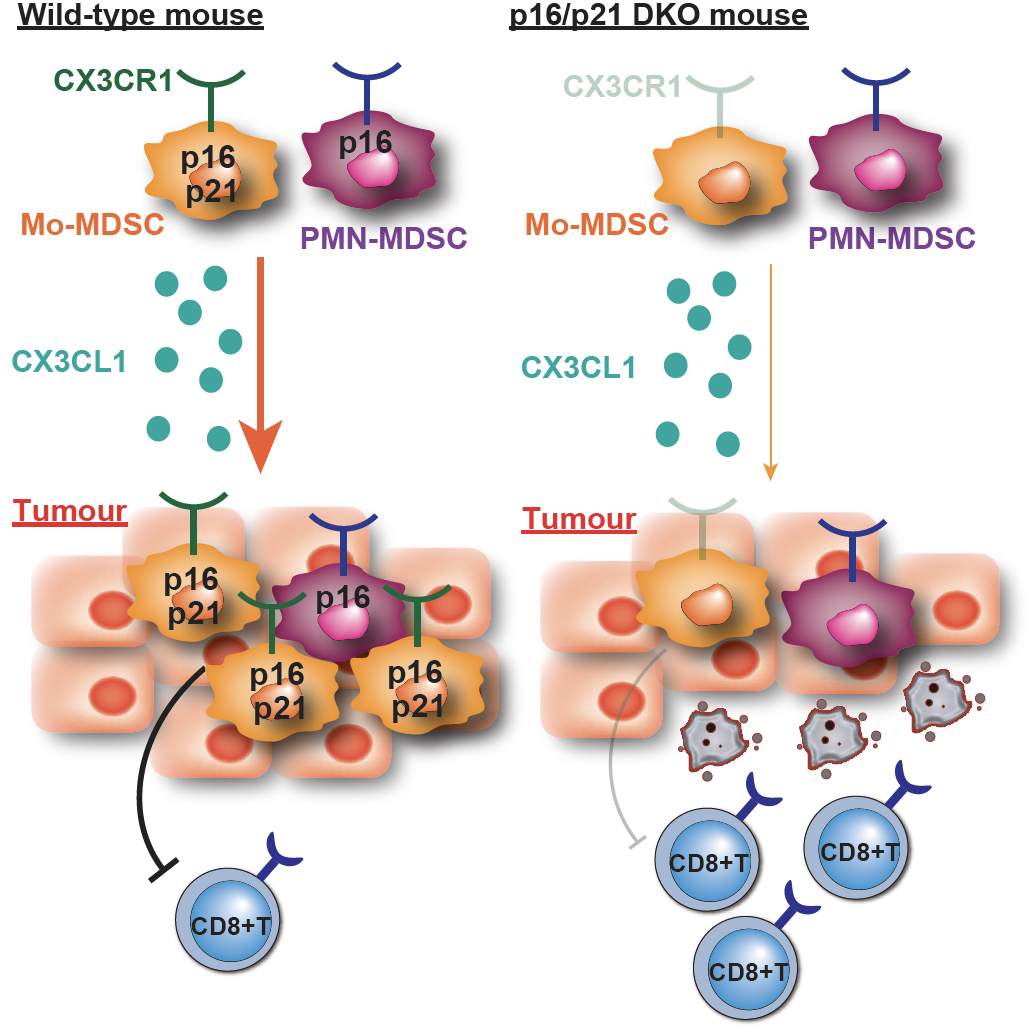
We reported an unexpected function of p16Ink4 and p21Cip1/Waf1, namely, tumour promotion through chemotaxis. In monocytic myeloid-derived suppressor cells (Mo-MDSCs) (*1), p16Ink4 and p21Cip1/Waf1 are highly expressed and stimulate CX3CR1 chemokine receptor expression by preventing CDK-mediated phosphorylation and inactivation of SMAD3 (Fig. 1). Accordingly, Mo-MDSC accumulation in tumours expressing CX3CL1 and the tumour progression in vivo are accelerated by p16Ink4 and p21Cip1/Waf1 (Fig. 2). Notably, blockade of the CX3CL1/CX3CR1-axis suppresses tumour growth, and pharmacological inactivation of CDKs elicits accelerated tumour growth.
These findings point out an unexpected function of p16Ink4a and p21Waf1/Cip1 and indicate that regulation of Mo-MDSC chemotaxis is a valuable potential strategy for control of cancer therapy.
*1 Mo-MDSC
A subtype of myeloid-derived suppressor cell (MDSC). MDSCs are known to negatively regulate immune responses and facilitate tumour progression.
Figure legends
(Upper Fig.) Molecular mechanism of p16/p21-mediated CX3CR1 expression in Mo-MDSCs and experimentally demonstrated effects of inhibition of the CX3CL1/CX3CR1 axis and CDK on tumour progression.
(Lower Fig.) Mo-MDSCs express both p16Ink4a and p21Cip1/Waf1, and migrate into CX3CL1-expressing tumours. However, loss of p16/p21 decreases CX3CR1-mediated infiltration of Mo-MDSCs into the tumour, resulting in attenuation of immunosuppressive ability in vivo.
This Article was published in Nature Communications on Dec 12, 2017.
p16Ink4a and p21Cip1/Waf1 promote tumour growth by enhancing myeloid-derived suppressor cells chemotaxis
- Home
- Achievement
- Research Activities
- Cellular senescence-associated factors in MDSCs promote tumour growth (Hara lab in Nat. Commun)