Suzuki Lab/Division of Host Defense Department of Immune Response Dynamics
We are studying the interactions between the nervous and immune systems with a special focus on how neural inputs control immune cell trafficking. Additionally, we are working on the development of novel therapies for inflammatory diseases.
- Immune regulation by the nervous system
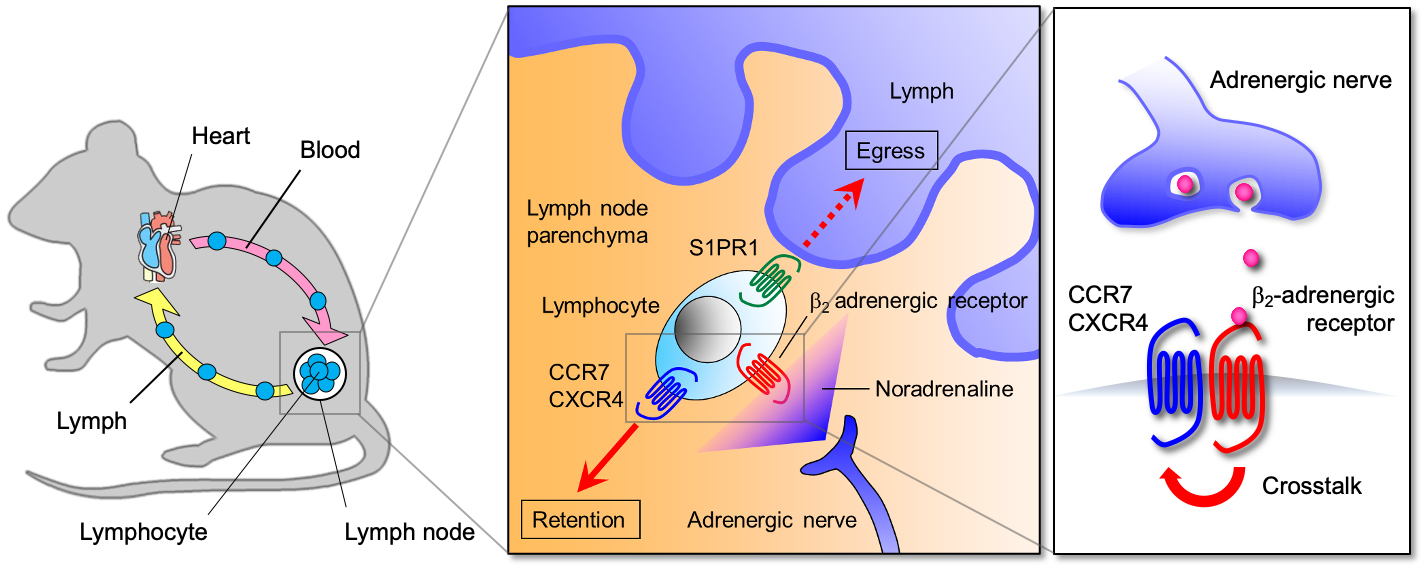
It has long been proposed that various aspects of immune responses are influenced by nervous system activity. Indeed, lymphoid organs are innervated by various types of neurons, and immune cells express neurotransmitter receptors to respond to the neural inputs. However, little is known about how the inputs from the nervous system control immune responses. To solve this problem, we are studying the cellular and molecular basis for the neural regulation of immunity. Adrenergic nerves constitute the efferent arc of the sympathetic nervous system and produce noradrenaline that induces cellular responses through α1-, α2-, β1-, β2- and β3-adrenergic receptors. Like other vital organs, lymphoid organs receive a rich supply of adrenergic nerves. We found that inputs from adrenergic nerves control lymphocyte egress from lymph nodes through β2-adrenergic receptors (Fig. 1). Additionally, our study demonstrated that this mechanism helps generate a diurnal rhythm of adaptive immune responses in lymph nodes.
- Developing novel therapeutic strategies for inflammatory diseases
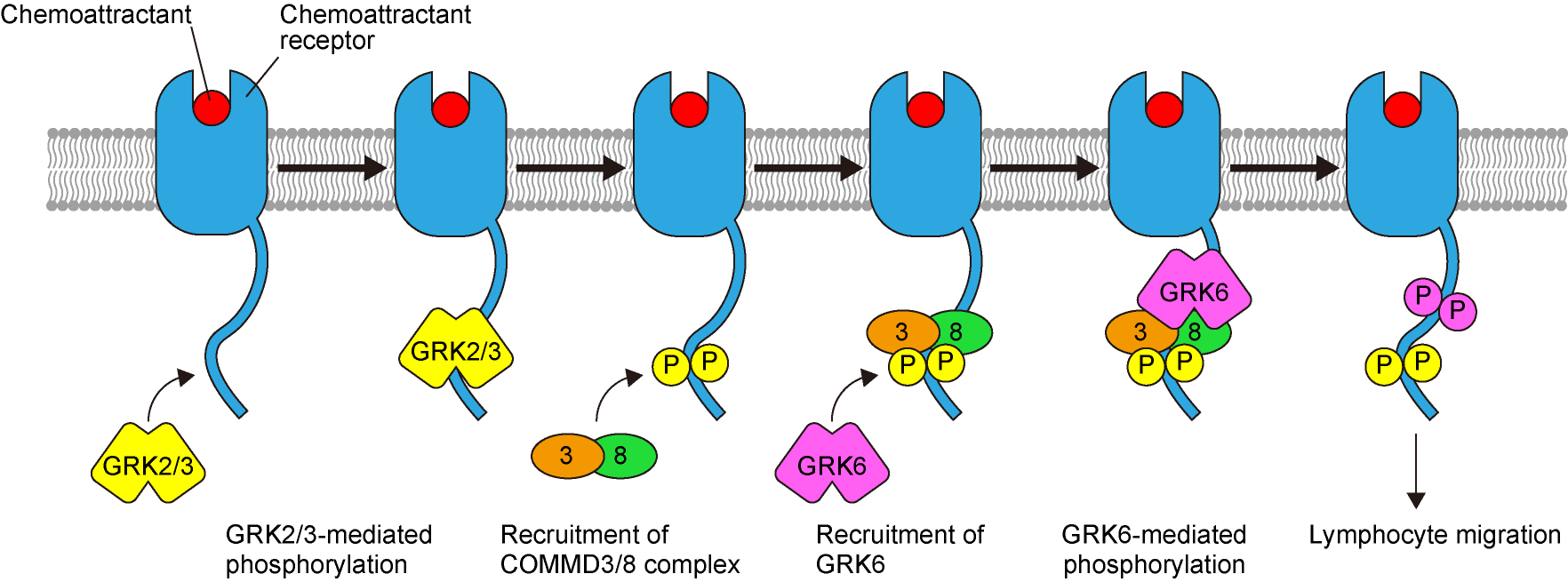
Lymphocyte migration is mediated by G protein-coupled receptors (GPCRs) that respond to chemoattractants, represented by chemokines. In search of novel factors involved in chemoattractant receptor signaling, we identified a protein complex consisting of copper metabolism MURR1 domain-containing (COMMD) 3 and COMMD8 (COMMD3/8 complex). The COMMD3/8 complex interacts with the C-terminal tail of chemoattractant receptors and promotes chemotaxis of lymphocytes (Fig. 2). We also found that the COMMD3/8 complex plays important roles in the control of B cell migration and induction of humoral immune responses. Our recent study suggested that the COMMD3/8 complex is involved in the pathogenesis of inflammatory diseases. Pharmacological inhibition of the COMMD3/8 complex may provide a novel approach for the treatment of immune disorders.
-
Fig. 1. Adrenergic control of lymphocyte egress from lymph nodes
Activation of β2-adrenergic receptors expressed on lymphocytes enhances the responsiveness of CCR7 and CXCR4, chemokine receptors that promote lymph node retention of lymphocytes, and inhibits their egress from lymph nodes.
-
Fig. 2. Role of the COMMD3/8 complex in chemoattractant receptor signaling
The COMMD3/8 complex functions as an adaptor that recruits GPCR kinase (GRK) 6 to chemoattractant receptors, promoting MAPK activation and consequently lymphocyte chemotaxis.
-
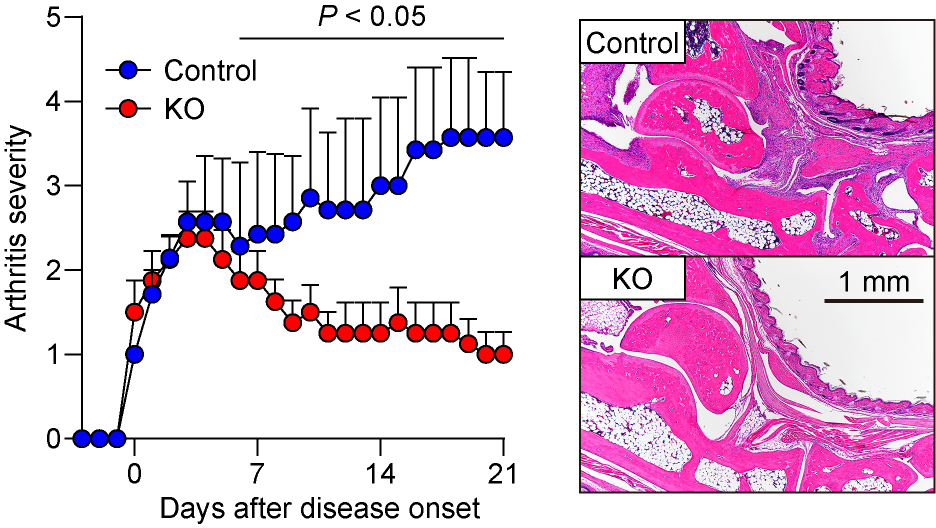
Fig. 3. Involvement of the COMMD3/8 complex in the pathogenesis of inflammatory diseases.
Deficiency of the COMMD3/8 complex (KO) inhibits the progression of collagen-induced arthritis, a mouse model of rheumatoid arthritis.
Staff
- Prof.: Kazuhiro Suzuki (concur.)
- Asst. Prof. : Taiichirou Shirai (concur.)
- Asst. Prof. : Satomi Komori
Website
Publications
- 1. Celastrol suppresses humoral immune responses and autoimmunity by targeting the COMMD3/8 complex. Shirai, T., et al., Sci. Immunol. (2023) 8: eadc9324.
2. The COMMD3/8 complex determines GRK6 specificity for chemoattractant receptors. Nakai, A., et al., J. Exp. Med. (2019) 216: 1630-1647.
3. Adrenergic control of the adaptive immune response by diurnal lymphocyte recirculation through lymph nodes. Suzuki, K., et al., J. Exp. Med. (2016) 213: 2567-2574.
4. Control of lymphocyte egress from lymph nodes through β2-adrenergic receptors. Nakai, A., et al., J. Exp. Med. (2014) 211: 2583-2598.
- Home
- Laboratories
- Suzuki Lab