Research achievements from Sato Lab published in Nat Commun.
M cells are specialized antigen-sampling cells that take up intestinal luminal antigens. A research group led by Specially-Appointed Associate Professor Shintaro Sato of Research Institute for Microbial Diseases at Osaka University and Professor Hiroshi Kiyono of the Institute of Medical Science at the University of Tokyo has identified that Allograft Inflammatory Factor 1 (Aif1) is an important functional molecule for bacterial and particle transcytosis function in M cells.
In the systemic immunity, antigen presentation is occurred in lymph nodes by antigen presenting cells (APCs), which capture antigens and then migrate to there through an afferent vessel. The antigen presentation of the mucosal immune system is occurred in mucosa associated lymphoid tissue (MALT); however, an afferent vessel is not present in MALT. Instead, the mucosal immune system has several antigen sampling systems directly from the mucosal surface. M cells are considered as the major player for antigen uptake. It has been reported that several receptors for microorganism are expressed on the apical surface of M cells and contribute to effective uptake of those microorganisms. However, the functional molecules, which are directly involved in the antigen transcytosis of M cells, are not yet reported.
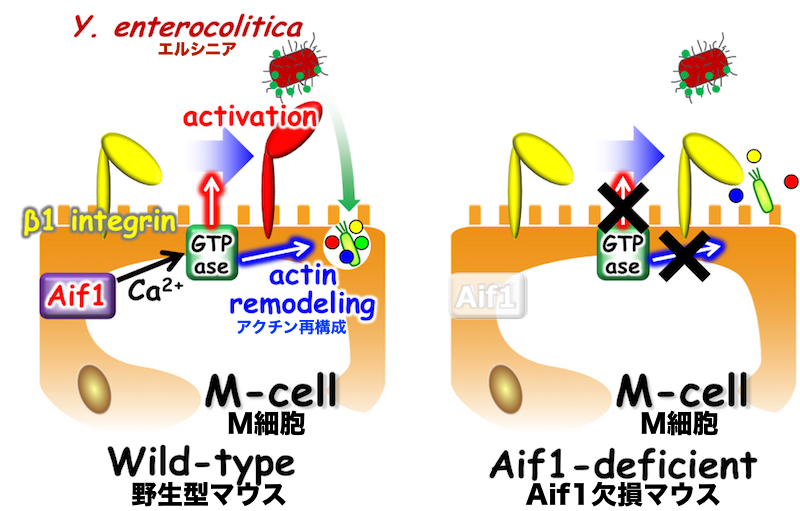
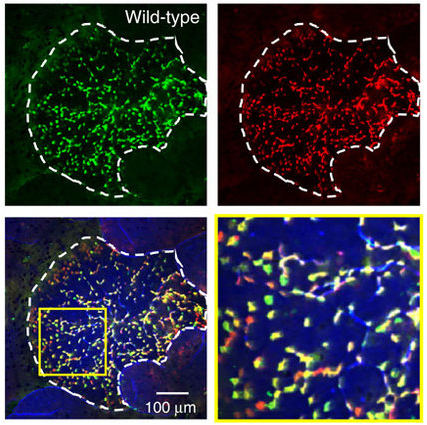
Here, the research group has shown that Aif1 is expressed by M cells and contributes to M-cell transcytosis by regulating actin remodeling. Aif1-deficient mice have shown suppressed uptake of particles and commensal bacteria, compared with wild-type mice. Translocation of Yersinia enterocolitica is also diminished in Aif1-deficient mice. Although β1 integrin, which acts as a receptor for Y. enterocolitica via invasin protein, is expressed on the apical surface membranes of M cells, its active form is rarely found in Aif1-deficient mice. These findings show that Aif1 is important for bacterial and particle transcytosis in M cells, and contribute to the development of more effective mucosal vaccine by increasing uptake of vaccine antigens and to the infection prophylaxis of pathogenic bacteria by reducing uptake of pathogen via M cells.
Figure legend
In wild-type mice, Aif1 is expressed in M cells. Aif1 can activate GTPase such as Rac in Ca2+-dependent manner. Activated GTPase induces actin remodeling at the apical surface of M cell leading to the antigen-uptake. GTPase also changes β1 integrin to its active form, which contributes to effective uptake of Yersinia enterocolitica via M cells. In Aif1-deficient mice, because GTPase is not able to be activated, both uptake of antigens and invasion of pathogenic bacteria are diminished.
Links
- Home
- Achievement
- Research Activities
- Research achievements from Sato Lab published in Nat Commun.