Kobayashi Lab/Research Center for Infectious Disease Control Department of Virology
Members of the family Reoviridae possess 9–12 segmented double-stranded RNA genomes. This family includes important pathogens, including the rotaviruses, in humans and animals. In our laboratory, using original technology to generate recombinant Reoviridae viruses from cloned cDNAs, we are studying molecular mechanisms underlying Reoviridae virus replication and pathogenesis, and developing novel vaccine vectors. More recently, we have also succeeded in developing a unique reverse genetics system using small fish for norovirus, which, like rotavirus, causes diarrheal disease. We are further extending this genetic manipulation approach to basic research on other diarrheal viruses, including norovirus.
1) Rotavirus
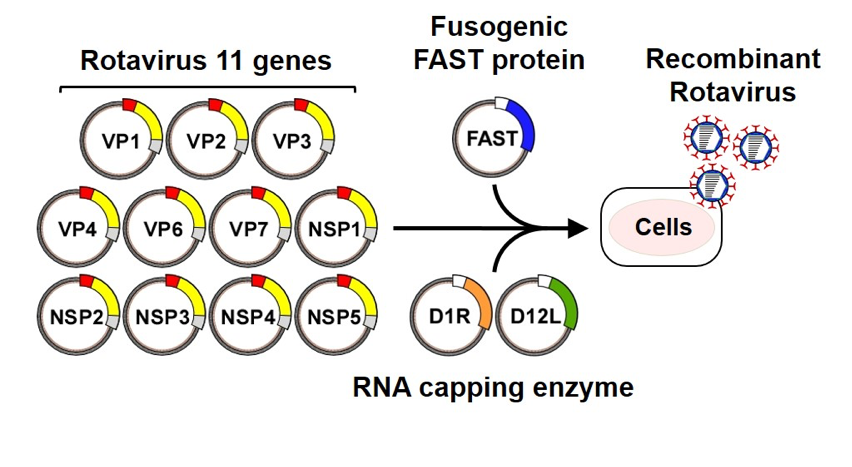
Rotaviruses (RVs) are highly important pathogens that cause severe diarrhea in infants and young children worldwide. Understanding of the molecular mechanisms underlying the replication and pathogenesis of RVs has been hampered by the lack of a reverse genetics system that allows the synthesis of recombinant viruses from artificial genes. In 2017, we developed a plasmid-based reverse genetics system for RVs. We are investigating RV biology and developing vaccines and therapeutics using a combination of genetic, biochemical, and biophysical approaches.
2) Norovirus
Noroviruses are highly important pathogens that cause acute gastroenteritis worldwide. Understanding of the molecular mechanisms underlying the replication and pathogenesis of noroviruses has been hampered by the lack of robust culture and genetic manipulation systems. Recently, we developed a long-awaited reverse genetics system for noroviruses. We are investigating norovirus biology and developing preventive and therapeutic strategies using this technology.
3) Oncolytic viral therapy using reovirus
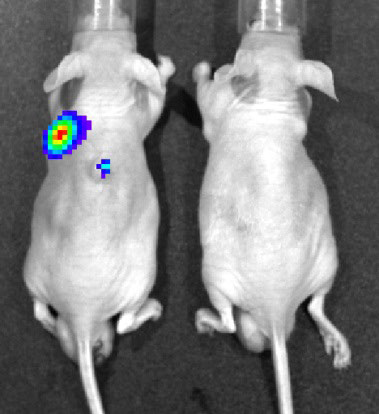
Mammalian orthoreoviruses (reoviruses) are highly tractable experimental models for studying the replication and pathogenesis of dsRNA viruses. Reoviruses have been evaluated as oncolytic agents against a variety of tumors, including head and neck, colon, breast, and pancreatic cancers, in animal models and humans. Wild-type reovirus-based oncolytic therapies are safe, but their efficacy is currently limited. We are developing safer and more effective reovirus-based cancer therapeutics by genetic modification.
4) Highly pathogenic bat-borne reovirus
Nelson Bay reovirus (NBV) was isolated from a flying fox in 1968 but had not been associated with any disease. However, NBVs were recently isolated from human patients suffering from acute respiratory tract infections in Malaysia, Indonesia, China, and Japan. These isolates have given rise to increasing concerns about bat-transmitted reovirus infections in humans. We are investigating how NBV replicates and causes disease in vitro and in vivo.
-
Fig.1. Generation of recombinant rotaviruses from cloned cDNAs. BHK-T7 cell lines were transfected with the rotavirus 11 cDNA constructs and expression plasmids encoding FAST and vaccinia virus capping enzyme (D1R and D12L).
-
Fig. 2. Generation of recombinant noroviruses from cloned cDNA. A norovirus infectious cDNA clone was microinjected into zebrafish embryos.
-
Fig. 3. Bioluminescence imaging of reovirus infection in human cancer xenografts. BALB/c nude mice transplanted with a human cancer cell line were infected intravenously with reporter reovirus.
Staff
- Prof.: Takeshi Kobayashi
- Asst. Prof.: Tomohiro Kotaki
- Asst. Prof.: Takahiro Kawagishi
- Sa Asst. Prof.: Shohei Minami
Website
Publications
(1) Recovery of infectious recombinant human norovirus using zebrafish embryos. Kotaki T., et al., PNAS. (2025)122:e2526726122.
(2) Single-round infectious rotaviruses with deletions of VP7 or VP4 genes, based on SA11 and WC3 strain backbones, and their potential use as viral vectors. Kotaki T., et al., PLoS Pathog. (2025)21:e1013484.
(3) A rotavirus VP4 or VP7 monoreassortant panel identifies genotypes that are less susceptible to neutralization by systemic antibodies induced by vaccination or natural infection. Kotaki T., et al., mBio. (2025)16:e0089725.
(4) Establishment of reverse genetics systems for Colorado tick fever virus. Minami S., et al., PLoS Pathog. (2025)21:e1012921.
(5) The nonstructural p17 protein of a fusogenic bat-borne reovirus regulates viral replication in virus species- and host-specific manners. Nouda R., et al., PLoS Pathog. (2022)18:e1010553.
(6) Development of an entirely plasmid-based reverse genetics system for 12-segmented double-stranded RNA viruses. Nouda R., et al., PNAS. (2021)118:e2105334118.
(7) Cell-cell fusion induced by reovirus FAST proteins enhances replication and pathogenicity of non-enveloped dsRNA viruses. Kanai Y., et al., PLoS Pathog. (2019)15:e1007675.
(8) Entirely plasmid-based reverse genetics system for rotaviruses. Kanai Y., et al., PNAS. (2017)114:2349-2354.
(9) Reverse Genetics for Fusogenic Bat-Borne Orthoreovirus Associated with Acute Respiratory Tract Infections in Humans: Role of Outer Capsid Protein sigmaC in Viral Replication and Pathogenesis. Kawagishi T., et al., PLoS Pathog. (2016)12:e1005455.
- Home
- Laboratories
- Kobayashi Lab