Reevaluation of the Efficacy of Senolytic Agents: The Importance of Reproducibility and Rigorous Evaluation (Hara Lab, in EMBO Reports)
A research group led by Professor Eiji Hara at the Research Institute for Microbial Diseases, The University of Osaka, together with Associate Professor Shimpei Kawamoto (currently Professor at Tohoku University), in collaboration with the group of Professor Yuichi Oike (Kumamoto University), the group of Dr. Akiko Takahashi (Cancer Institute, Japanese Foundation for Cancer Research), the group of Associate Professor Hiroshi Kondoh (Kyoto University), the group of Dr. Masataka Sugimoto (National Center for Geriatrics and Gerontology), the group of Professor Michiyuki Matsuda (Kyoto University), and the group of Dr. Naoki Mochizuki (National Cerebral and Cardiovascular Center) conducted independent and collaborative cross-laboratory validation of previously reported senolytic agents. Their findings challenge earlier claims and emphasize the critical importance of reproducibility and methodological rigor in senolytic research.
Cellular senescence is a stable cell-cycle arrest mechanism that plays an essential role in tumor suppression. However, senescent cells accumulate with age and secrete pro-inflammatory factors known as the senescence-associated secretory phenotype (SASP), which contributes to age-related diseases. Consequently, senolytic drugs—agents that selectively eliminate senescent cells—have attracted considerable attention as a promising therapeutic strategy to extend healthspan. However, given the strong societal interest in this field, especially careful and rigorous evaluation is required.
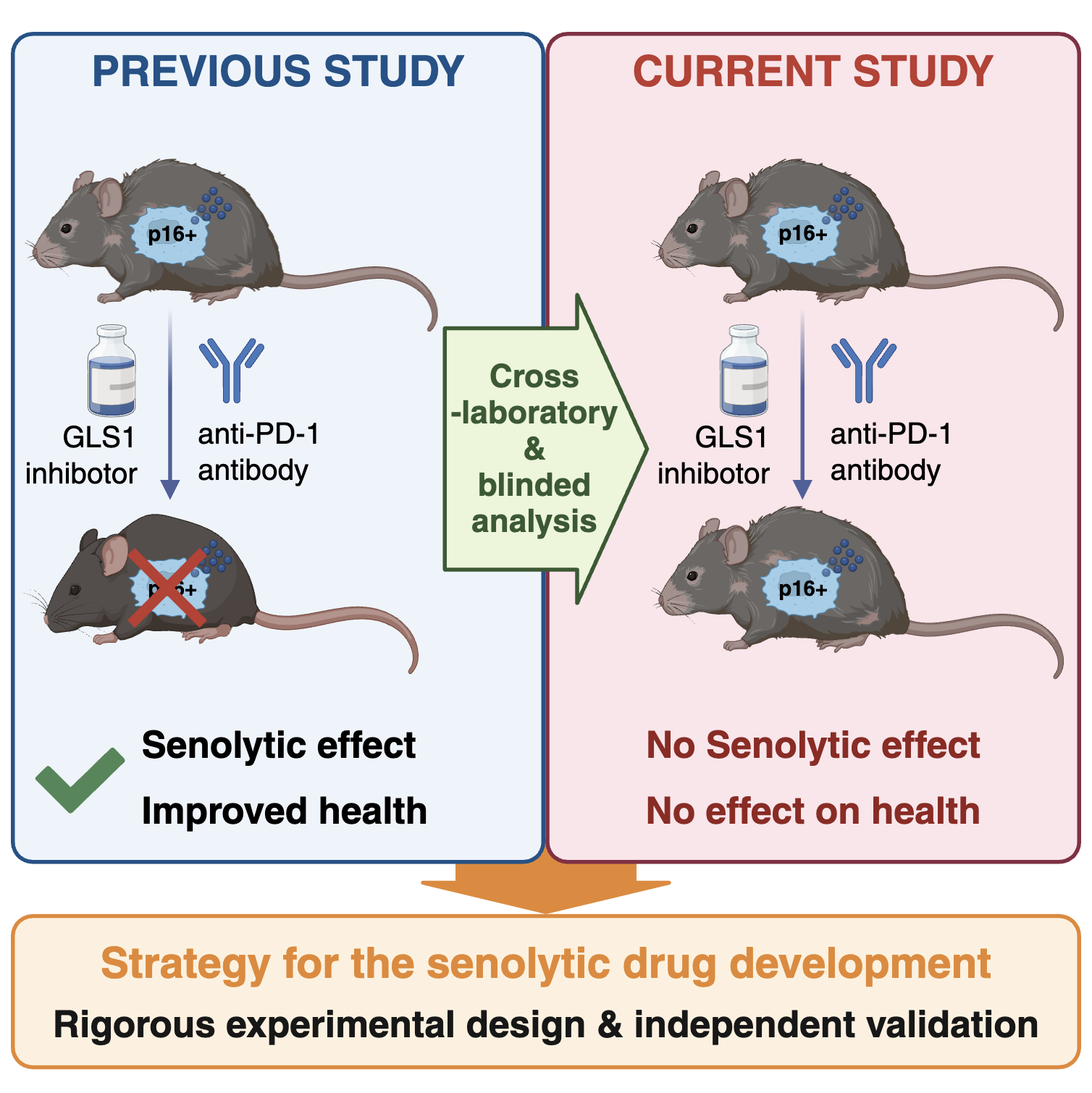
In this study, the researchers rigorously reexamined the senolytic efficacy of a GLS1 inhibitor (BPTES) and an anti–PD-1 antibody, both of which had previously been reported to reduce senescent cell burden in aged mice. Using blinded and cross-institutional experimental designs, they found that neither treatment significantly reduced endogenous p16INK4a expression in multiple organs, including the liver, lung, and kidney, nor improved aging-related phenotypes, including external appearance and functional measures such as grip strength. In addition, in vitro analyses independently conducted by multiple laboratories revealed that BPTES affected not only senescent cells but also non-senescent cells, suggesting limited selectivity as a senolytic agent.
A key strength of this study lies in its rigorous design, including independent replication across multiple laboratories and blinded analysis to minimize experimental bias. These approaches are particularly important given the high inter-individual variability observed in aged animals.
While the present findings do not necessarily invalidate previous studies, they underscore the need for cautious interpretation and highlight the importance of standardized, reproducible methodologies in the development of senolytic agents. This work provides a critical foundation for advancing more reliable and translatable approaches targeting cellular senescence.
This study was published online in EMBO Reports on April 3, 2026.
Title: Reevaluating the Senolytic Activity of a GLS1 Inhibitor and an Anti-PD-1 Antibody: Toward Greater Reproducibility and Methodological Rigor
Authors: Shimpei Kawamoto, Haruki Horiguchi, Daisuke Torigoe, Masahiro Wakita, Koyu Ito, Sho Sugawara, Xiangyu Zhou, Takumi Mikawa, Jeong Hoon Park, Birte Kristin Jung, Yumiko Okumura, Hideka Miyagawa, Mikako Maruya, Nozomi Hori, Ken Uemura, Masataka Sugimoto, Michiyuki Matsuda, Naoki Mochizuki, Hiroshi Kondoh, Akiko Takahashi, Yuichi Oike, & Eiji Hara
DOI: 10.1038/s44319-026-00740-5
Links
- Home
- Achievement
- Research Activities
- Reevaluation of the Efficacy of Senolytic Agents: The Importance of Reproducibility and Rigorous Evaluation (Hara Lab, in EMBO Reports)